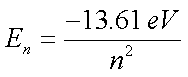

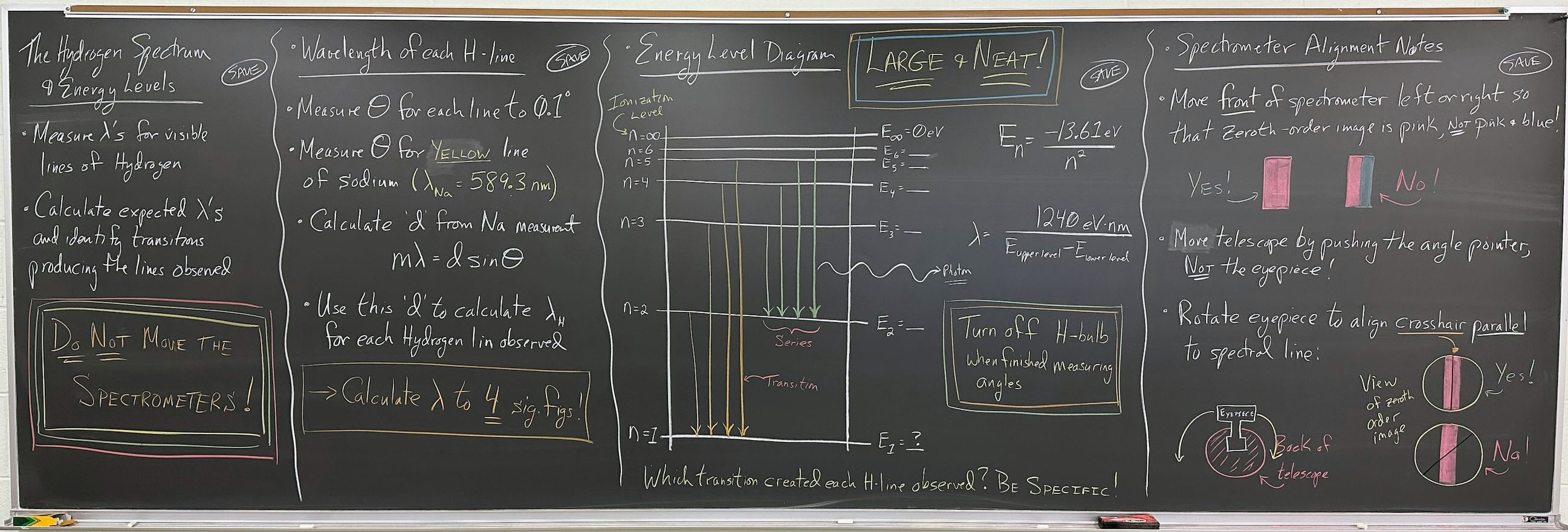
The Hydrogen Spectrum & Energy Levels
BlackBoard Notes
======================= Photo - Click for a Larger Version =============================
======================= First Board Section =============================
- Measure λ's for visible lines of Hydrogen
- Calculate expected λ's and identify transitions producing the lines observed
- Do Not Move the Spectrometers!
- (If needed:) Lab Evaluations!
- On front bench
- Fill one in (out?)
- No name required
- Leave pages stapled together
======================= Second Board Section =============================
- Wavelength of each H-line:
- Measure θ for each line, to 0.1°
- Measure θ for Yellow line of sodium (λNa
= 589.3 nm)
- Calculate d from Na measurement:
- Use this d to calculate λH for each hydrogen line observed
- Calculate λ to 4 sig figs!
======================= Third Board Section =============================
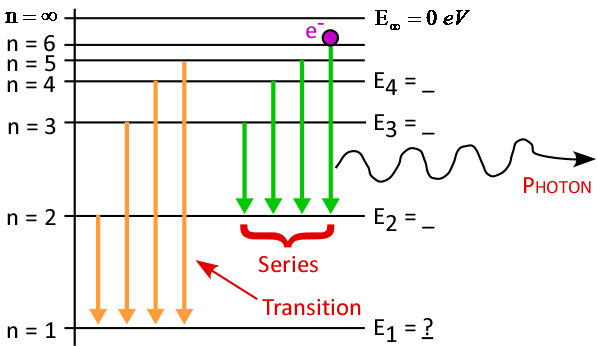
- Energy level diagram (Large and Neat!):
- Drawing note for Jeff:
- First draw horizontal lines for n = ∞ and n = 1
- Label n = ∞ as "Ionization Level"
- Fill in other levels by taking ½ of available space
- Finish with vertical axis and transitions
- Equations may run into the Fourth Board Section
- Which transitions created each H-line observed? Be specific!
- Turn off the H-bulb when finished measuring your angles!
======================= Fourth Board Section =============================
Spectrometer Alignment Notes: (Note that this section will be tight on space!)
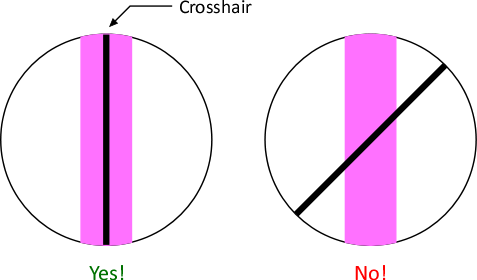
- Move front of spectrometer left and right so zeroth order image is pink, not pink and blue!
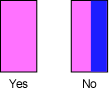
- Move telescope by pushing the angle pointer, NOT the eyepiece!
- Rotate eyepiece to align crosshair parallel to spectral line.
Return to Setup
|
Revised: 12 Jun 24
|
Canton, NY 13617
|